 The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore. In Chapter 1, we described Dalton’s theory that each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. Obtain the atomic masses of each element from the periodic table and multiply the atomic mass of each element by the number of atoms of that element. When using the unit g/mol, the numerical value of the molar mass of a molecule is the same as its average mass in. The molar mass has units of g/mol or kg/mol. A mole of substance is equal to Avogadros number (6.023×10 23) of that substance. See original paper for the range of these elements from different sources Isotope-abundance variations and atomic weights of selected elements: 2016 (IUPAC Technical Report), Pure Appl. The molar mass is the mass of one mole of substance, whether the substance is an element or a compound. A property closely related to an atom’s mass number is its atomic mass. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number. 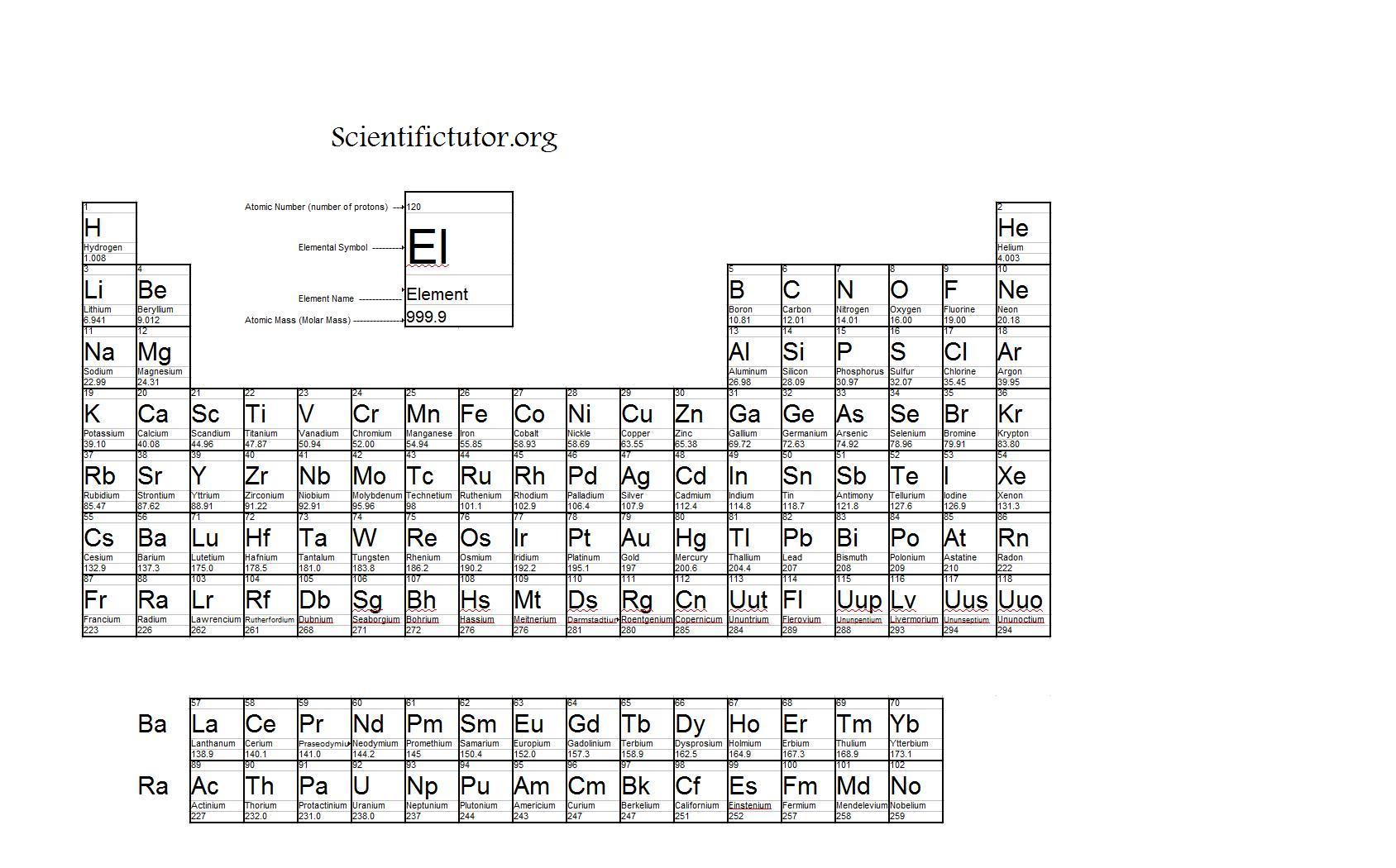
(2 atoms)(30.973761 amu/atom) = 6.0474 amu Using a mass spectrometer, a scientist determined the percent abundances of the isotopes of sulfur to be 95.27 for 32 S, 0.51 for 33 S, and 4.22 for 34 S. List of Elements with Range of Atomic Weights. Together, the number of protons and the number of neutrons determine an element’s mass number: mass number protons + neutrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
